A TIMELY REMINDER…
Was it REALLY safe and effective?
QUESTION: HOW, on 14th September 2020, did the UK Medicines and Healthcare Products Agency (MHRA) actually KNOW there would be an “expected high volume of Covid-19 vaccine Adverse Drug Reaction (ADRs)”?
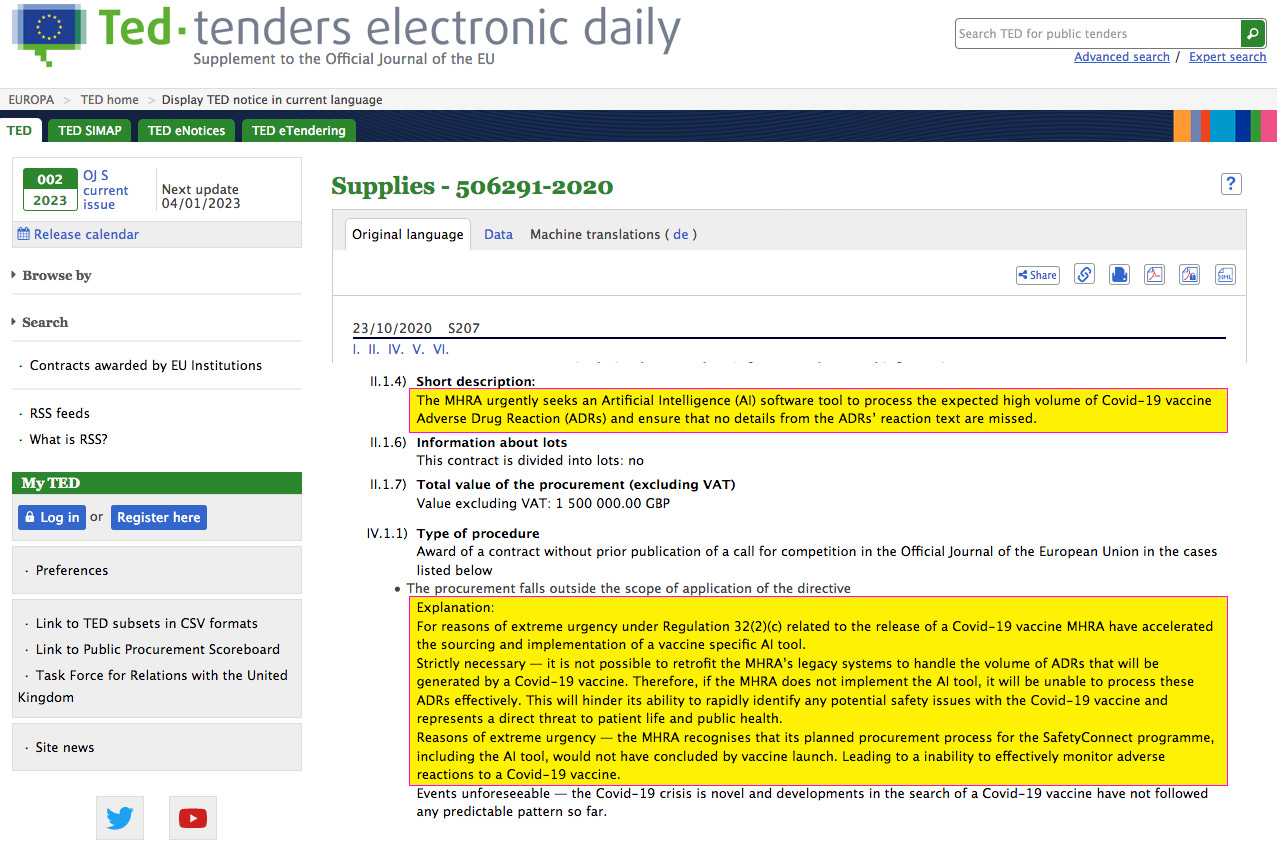
The MHRA urgently seeks an Artificial Intelligence (AI) software tool to process the expected high volume of Covid-19 vaccine Adverse Drug Reaction (ADRs)
The MHRA subsequently awarded a £1.5million Tender to Genpact UK Ltd, with a requirement for an “Artificial Intelligence (AI) software tool to process the expected high volume of Covid-19 vaccine Adverse Drug Reaction (ADRs) and ensure that no details from the ADRs’ reaction text are missed”. REF WEB LINK 01
The MHRA states:
“For reasons of extreme urgency under Regulation 32(2)(c) related to the release of a Covid-19 vaccine MHRA have accelerated the sourcing and implementation of a vaccine specific AI tool. Strictly necessary — it is not possible to retrofit the MHRA’s legacy systems to handle the volume of ADRs that will be generated by a Covid-19 vaccine. Therefore, if the MHRA does not implement the AI tool, it will be unable to process these ADRs effectively. This will hinder its ability to rapidly identify any potential safety issues with the Covid-19 vaccine and represents a direct threat to patient life and public health.”
Janet Menage wrote in a BMJ Rapid Response:
“Why do they expect that a high volume of ADRs will be generated? And would it not be more sensible to determine the safety profile of a Covid-19 vaccine while the stable door is shut, rather than monitor the galloping horse once it has bolted?” REF WEB LINK 02
I ask again:
HOW did the MHRA know there would be an expected high volume of Vaccine Adverse Drug Reactions?
Max Hogster
January 2023
FOR REFERENCE:
UK Government Vaccine Adverse Reaction stats:
https://yellowcard.ukcolumn.org/yellow-card-reports


esp